Collaboration between patient organisations and a clinical research sponsor in a rare disease condition: learnings from a community advisory board and best practice for future collaborations | BMJ Open
Clinical Trial Data Management and Electronic Recruitment Solutions: Impactful Ways to Promote Successful Clinical Trials Using

Clinical Study Card Programs, Patient Assistance, Co-Pay, & Clinical Study Programs From RxSolutions, Including … | Study cards, Free trial offer, Study program

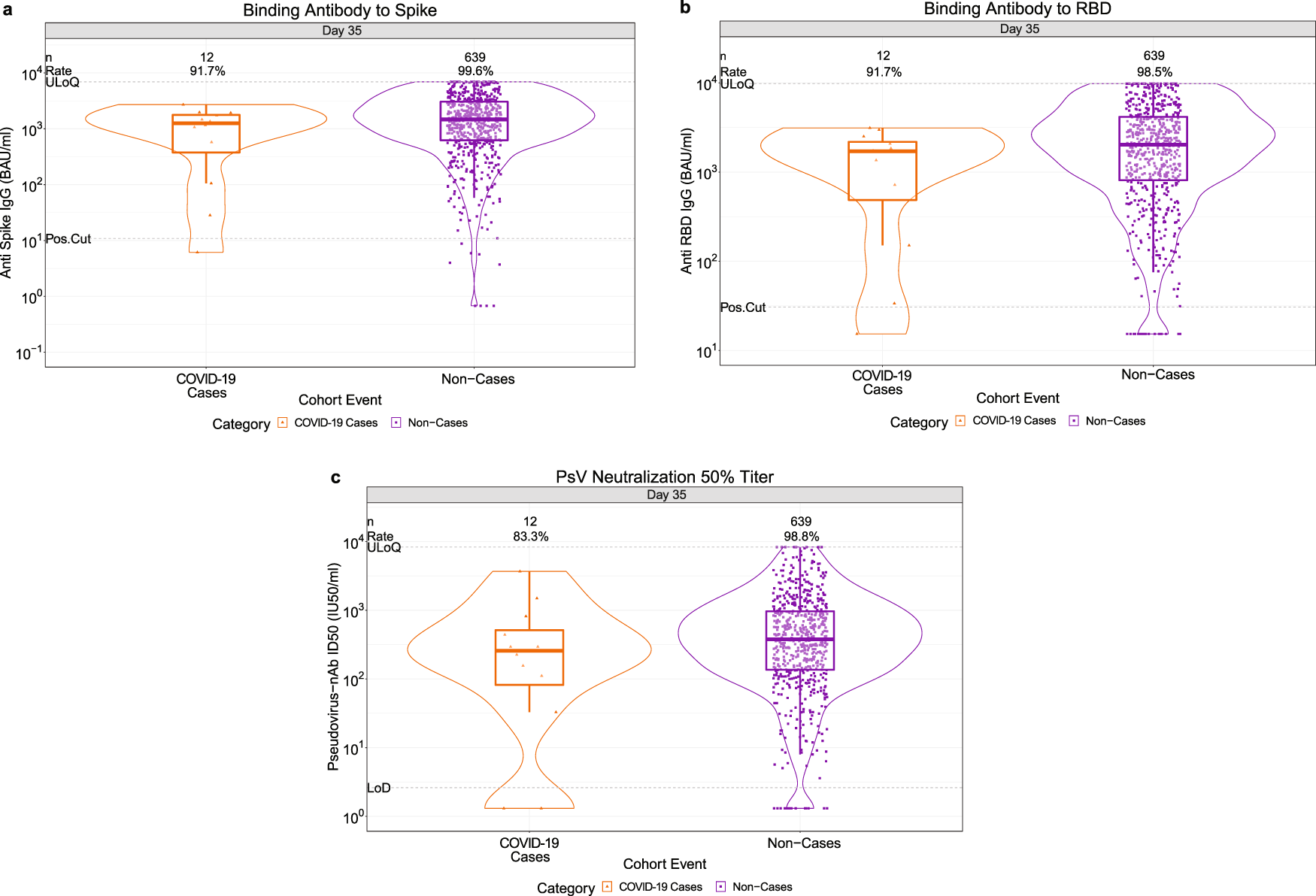
Evaluation of safety and immunogenicity of receptor-binding domain-based COVID-19 vaccine (Corbevax) to select the optimum formulation in open-label, multicentre, and randomised phase-1/2 and phase-2 clinical trials - eBioMedicine
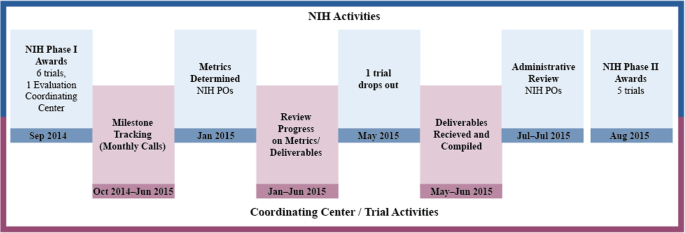
Is this study feasible? Facilitating management of pragmatic trial planning milestones under a phased award funding mechanism | Trials | Full Text
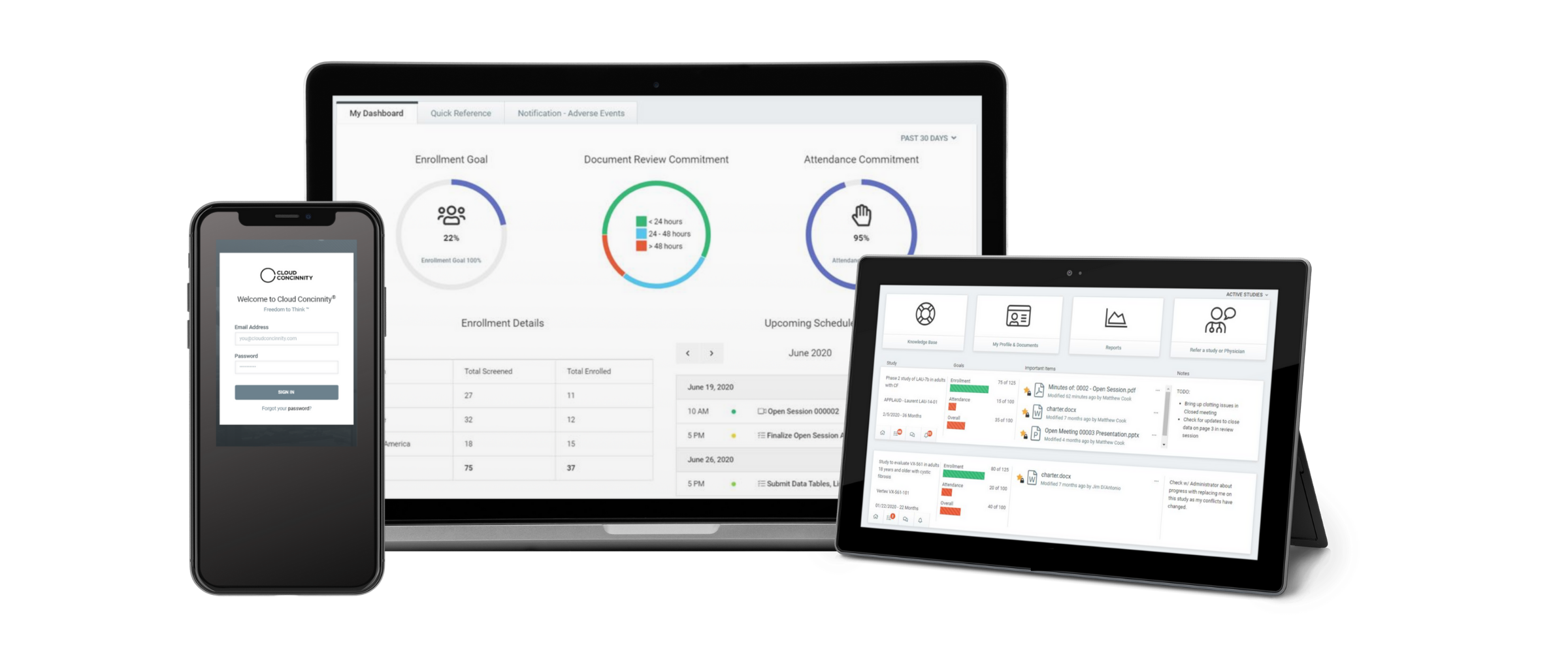
Clinical Trial Oversight | DSMB Software | Data Monitoring Committee in Clinical Trials | Cloud Concinnity®

Safety and efficacy of the mRNA BNT162b2 vaccine against SARS-CoV-2 in five groups of immunocompromised patients and healthy controls in a prospective open-label clinical trial - eBioMedicine